AMW GmbH
About us
AMW GmbH is a specialty pharmaceutical company focused on biodegradable implants, providing patients and partners access to value-added medicines for improved adherence and cost-effective therapies. The Company covers the entire value chain, from (co-)development to manufacturing and international commercialization.
AMW has two approved medical products, implants releasing goserelin and leuprorelin, which are distributed worldwide through both local and global pharma partners. Based on its unparalleled expertise in formulation development, the Company collaborates with pharma and biotech partners worldwide to develop and manufacture high-quality products and life-cycle solutions.
Address
Birkerfeld 11
83627 Warngau
Germany
E-mail: l.grundbacher@amw-pharmaceuticals.com
Phone: +49 8024 470999-0
Internet: amw-pharmaceuticals.com/en/
Contact person:
Dr. Joao Silva
E-mail: j.silva@amw-pharmaceuticals.com
Products & Services
CDMO+
Contract development and manufacturing
We offer appropriate service models for partners who are looking for expertise in the development and manufacturing of biodegradable implant formulations. Following process transfer, complete and certified upscaling is ensured based on GMP manufacturing units and quality control procedures.
Contract manufacturing
The entire value stream is controlled within AMW, from logistics to manufacturing and shipping. AMW has many years of expertise in implant manufacturing using extrusion technology in a GMP-regulated environment. In melt extrusion, conical twin-screw extruders are used to mix active ingredient molecules with polymers. The focus is on the state-of-the-art in melt extrusion as well as the hygienic requirements according to Annex 1 of the EU-GMP guidelines.
Primary packaging is in syringes/applicators. The packing area allows us to pack a wide variety of formats while complying with serialization and track trace (including aggregation) specifications. AMW attaches great importance to a sustainable quality assurance process that begins with the selection of suppliers and extends through incoming goods inspection of the raw materials, packaging materials, in-process control, and the release of the finished goods for shipment or the market at the end of the production chain.
Our employees with their many years of experience in the GMP-regulated environment lay the groundwork for these processes.
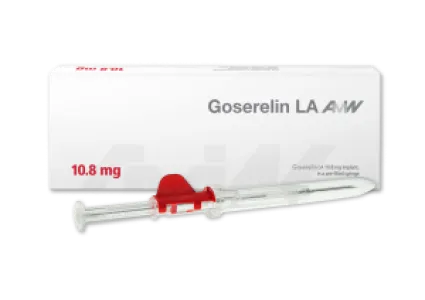
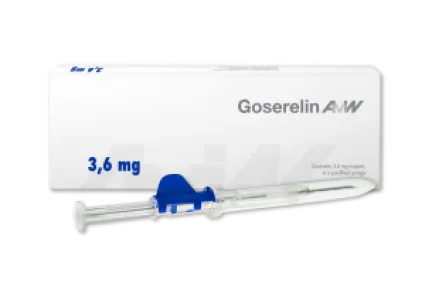
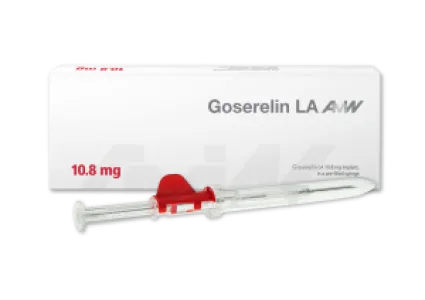
Goserelin - one (3.6 mg dosage) or three (10.8 mg dosage) months
The active ingredient goserelin is a synthetic peptide that can be used primarily in the treatment of hormone-dependent tumors, particularly prostate cancer. Thus, goserelin intake has indirect effects on testosterone levels in men, the lowering of which stops the growth of malignant cancerous tumors. In addition, goserelin implant is used to treat certain benign gynecological disorders. Goserelin is administered in the form of a biodegradable implant that releases the active ingredient over a period of either one (3.6 mg dosage) or three (10.8 mg dosage) months. Goserelin is the only generic of the originator with global availability.
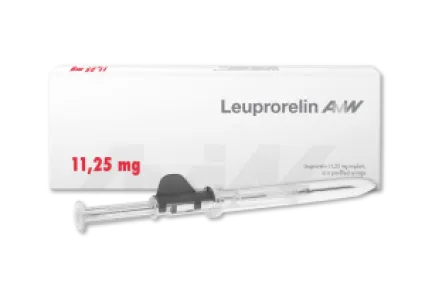
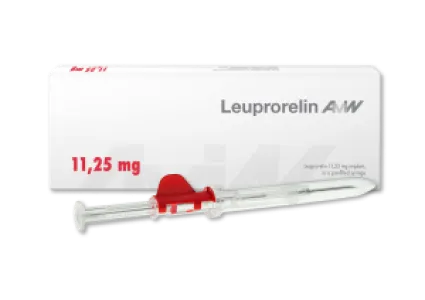
Leuprorelin - three-month (11.25 mg dosage)
The active ingredient leuprorelin, like goserelin, is a synthetic peptide that can be used primarily in the treatment of hormone-dependent tumors, particularly prostate cancer. Administration of leuprorelin has indirect effects on testosterone levels in men, the lowering of which stops the growth of malignant cancerous tumors. Leuprorelin is administered in the form of a biodegradable implant that releases the active ingredient over a three-month period (11.25 mg dosage). Leuprorelin is the only implant formulation with a self-retracting needle, which makes administration much more convenient for patients.