PlasmidFactory GmbH
About us
PlasmidFactory GmbH, established in 2000 in Bielefeld, Germany, is a premier contract development and manufacturing organization specializing in plasmid and minicircle DNA. We support clients and partners in cancer research, gene and cell therapy, and vaccination, with a focus on CAR-T cell development, AAV gene transfer, and minicircle tools for mRNA synthesis.
We offer custom plasmid and minicircle DNA manufacturing services across various quality grades from Research and CCC grade up to High Quality grade and GMP certified material for clinical trials and applications. Our state-of-the-art facilities fulfil the highest quality standards, with full GMP services.
Our extensive In-Stock product range supports immediate research needs, including AAV production plasmids and reporter constructs. At the forefront of Next Generation Gene Vectors, our R&D focuses on advancing our proprietary minicircle DNA technologies. Through strategic collaborations with global partners, we are committed to pioneering solutions for academia and the biopharmaceutical industry.
- 25 years CDMO experience and innovation in plasmid and minicircle DNA
- +3500 successful projects
- From research grade to GMP
- Talk to us to discuss how we can support your projects in the area of CGT, AAV or mRNA
Address
Meisenstr. 96
33607 Bielefeld
Germany
E-mail: info@plasmidfactory.com
Phone: +49 521 2997 350
Internet: www.plasmidfactory.com/
Contact person:
Dr. Marcello Stein
Senior Director Commercial
E-mail: marcello.stein@plasmidfactory.com
Products & Services
Besides custom DNA manufacturing our portfolio includes different services such as GMP-compliant storage or the CGE (Capillary Gelelectrophoresis)-based analysis of plasmid topologies.
Large-scale plasmid DNA production in our modern facilities.
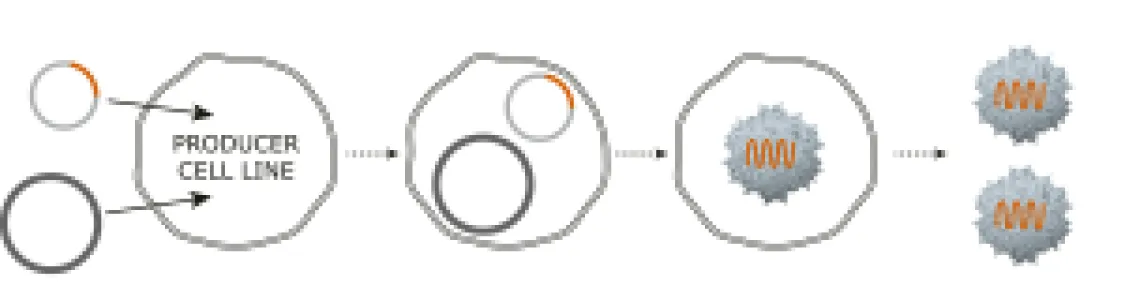
2-plasmid transfection system for AAV production.
Our second HQ facility: in operation since 2020 for large scale production of High Quality Grade DNA to be used as starting material in clinical trials.
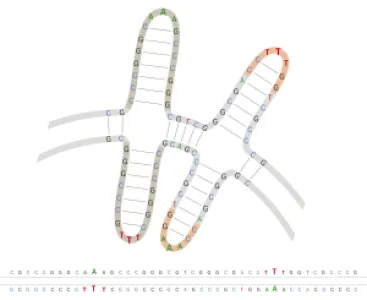
PlasmidFactory’s proprietary ITRRESCUE® technology enables amplification of plasmid DNA containing sensitive inverted terminal repeats (ITR).
Our new, state-of-the-art GMP plant, completed in 2025.




-thumb.webp)